A. S. Rossomagina
JCS Perm Production of Foamed Silicates
D. V. Saulin
I. S. Puzanov
Perm State Technical University
Russia
International Conference
University of Dundee 2008
ABSTRACT. The main obstacle for utilization of glass cullet as an aggregate in concrete is that it contains active amorphous silica which is highly reactive. So far, research workers have faced certain difficulties trying to use glass cullet and foamed glass for the production of binding materials or concrete fillers due to the reaction between the alkali contained in concrete (or in a filler) and the silica contained in glass (hereinafter ASR – alkali-silica reaction). The purpose of the research was to find a way of producing ASR-stable glass concrete. An effect of using highly dispersed silicate additives and an influence of chemical treatment of the glass aggregate have been studied. It was shown that glass as an aggregate can be used, with certain precautions, to manufacture a wide range of concrete products with no serious detrimental effects on a product performance.
Keywords: Alkali-Silica Reaction, Glass Aggregate Concrete.
A S Rossomagina is a consulting engineer at the JSC Perm Production of Foamed Silicates specializing in reuse of glass cullet in construction materials.
Dr. D V Saulin is a senior lecturer at the Chemical Engineering Department of the Perm State Technical University. Research interests include chemical engineering and simulation processes.
Dr. I S Puzanov is a senior lecturer at the Chemical Engineering Department of the Perm State Technical University. Research interests include reuse and recycling processes of waste materials.
INTRODUCTION
A number of attempts have been made to successfully use glass as an additive in concrete compositions.
Glass and glass-containing materials (glass wool, glass fibre cloth and foamed glass) have a number of valuable service properties, which make them very effective aggregates for concrete: high hardness, chemical resistance, availability and a relatively low cost.
But despite the apparent simplicity and evidence of such a solution, substituting waste glass and other glass-containing materials for natural aggregates in concrete has so far been unsuccessful because of the problem of the reaction between the alkali contained in concrete (or in aggregate) and the silica contained in glass (hereinafter ASR - alkali-silica reaction).
The reaction starts with the interaction between the alkali hydroxides produced from alkalis (Na2O and K2O) and the siliceous minerals of an aggregate. As a result, a gel-like substance forms composed of alkali silicates. Initially, the gel forms on the aggregate, then some gel can form within the aggregate as well, if it is porous. This gel is prone to swelling. It absorbs water and the volume of the gel increases. Under confinement by the cement matrix and aggregate, the swelling of the ASR gel generates hydrostatic pressure. If the reaction continues and the internal pressure exceeds the tensile strength of the matrix, cracks will form around the reactive aggregate particles. According to [1], the pressure can exceed 2 N/mm2, creating tensile forces in the concrete matrix which cause cracking of the concrete.
ASR occurs in ordinary concrete if a “wrong” kind of the aggregate, containing large amount of amorphous silica, is used. In a glass aggregate, ASR occurs with a great certainty – due to the fact that glass contains both amorphous silica and Na+ ions, contributing to a general alkalinity of the cement composition. Besides, the chemistry of soda-lime glass (used for common consumer containers) is quite simple compared with that of a natural aggregate. That makes glass almost an ideal aggregate with which one can study the ASR phenomenon and search for the methods to prevent it or to mitigate its effects, and with which one can perform parameter studies.
A number of authors have studied the methods of mitigating the ASR process which takes place in a production of glass concrete – concrete with a glass aggregate.
A serious research work has been carried out in the Columbian University (US) by Professor C. Meyer [2,3]. He proved that addition of glass into a composition of concrete causes alkali-silica reaction which results in a decrease of the concrete strength. M.Sugiyama [4] studied the influence of temperature and glass composition on the ASR process.
W. Remarque, D.Heinz, and C. Schleusser [5] discovered that the addition of a highly dispersed glass powder into the composition of concrete does not cause the ASR process. The authors suppose that in this case alkali-silica reaction proceeds at a high rate and the process terminates in 1-2 days. After this period of time, expansion of concrete blocks does not take place. Summarizing the available data, we can conclude that the possible ways of suppressing ASR in glass concrete are: use of glass with a certain granulometric composition [6,7], addition of highly dispersed glass [8], addition of lithium and zirconium compounds into the composition [2].
Therefore, our aim was to find a way of producing ASR-stable glass concrete. We have studied the effect of using supplementary cementing materials and the influence of chemical treatment of a glass aggregate.
EXPERIMENTAL DETAILS
The experiments were carried out according to the modified ASTM standard C1293-01. The concrete blocks were exposed to 60oC and 100% humidity for three months. The expansion of samples was measured every 14 days by an optical dilatometer. The toughness of samples was measured by a compression-testing machine IP 6010-100-1.
We used standard cement M400 produced by the Pashijsky cement plant.
Only chemically pure reagents were utilized for the experiments. Glass cullet was obtained from the mixture of window and bottle glass taken in the proportion 1:1. It was ground in a vibro-centrifugal mill.
The foamed glass aggregate used was produced by the Penosytal JSC.
RESULTS AND DISCUSSION
Most authors agree that there are two necessary conditions for ASR to take place in a cement composition – the presence of alkali (mostly in the form of NaOH) and the presence of reactive, usually amorphous, silica. In a glass aggregate, ASR occurs with a great certainty – due to the fact that glass contains both amorphous silica and Na+ ions, contributing to the general alkalinity of a cement composition.
So, there are two possible ways of ASR suppression: 1) binding of the excess of alkali (for example, with highly dispersed silicates), 2) transformation of silica on the surface into a less reactive form. Both methods have been investigated. Besides, the influence of granulometric composition of a glass aggregate on the ASR rate has been studied. In some cases the additional alkali was added into the composition.
Size of Glass Aggregate
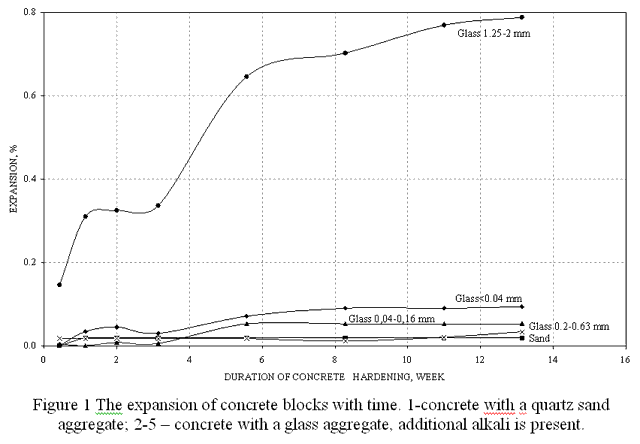
To evaluate the rate of alkali-silica reaction, a series of experiments has been conducted. The reaction rate was characterized by an expansion of sample concrete blocks and a decrease of their strength characteristics. Concrete with quartz sand aggregate was taken for reference. It was found out that a considerable expansion is observed only when additional alkali is added into the composition, in the case of a large particle size of the glass aggregate – more than 1.25 mm (Figure 1).
The analysis of strength characteristics of concrete blocks allowed us to reveal an interesting regularity. When the particle size of the aggregate is less than 0.05 mm (50 micrometers), unusual growth of the glass concrete block strength, exceeding the strength of the concrete on quartz sand, is observed. It can be explained by the ability of a highly dispersed glass to participate in the processes of forming new phases due to its high specific surface area.
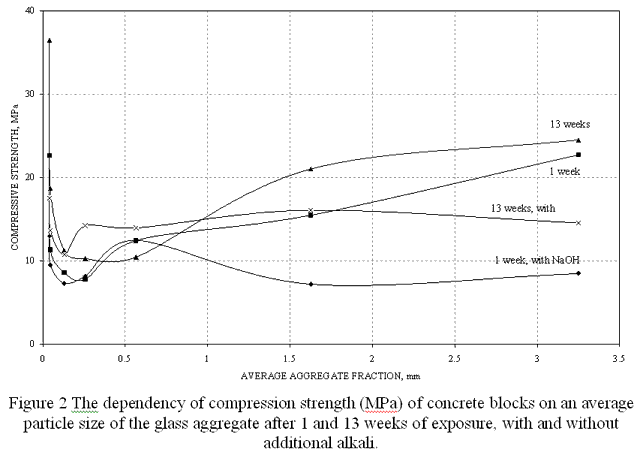
It should be also noted, that strength characteristics of alkali-free concrete samples are anomalously high – both for the minimum and maximum particle sizes of the aggregate. The corresponding dependencies are given on Figure 2.
All the curves have a distinct minimum, corresponding to the fraction of 0.1 – 0.3 mm. The behavior of the curves does not change – in the case of an alkali-free concrete, there is a steep ascent of the compression strength with the decrease of an average particle size and a slow ascent of the compression strength with the increase of an average particle size, and in the case of additional alkali–containing concrete compositions, the compression strength slightly grows and then stabilizes with the increase of an average particle size.
So, the use of glass cullet with a large particle size exceeding 1.2 mm can be recommended as an aggregate for concrete, when cement has a low alkalinity. The compression strength of such glass concrete exceeds the compression strength of common concrete with a quartz sand aggregate.
But the problem of alkali-silica reaction cannot be solved that way. First, the presence of free alkali in cement or in other components of concrete will cause a decrease of strength characteristics of concrete. Second, in the process of large-scale grinding of glass it is hard to prevent spontaneous crushing of large glass particles which also lowers the quality of the concrete.
So, we come to the conclusion, that the concrete with a glass aggregate of a medium particle size, typical for real-life environment, is susceptible to ASR. Then, it is necessary to take additional measures to prevent ASR in case of utilizing common glass cullet as an aggregate for concrete.
Dispersed Silicate Additives
It was shown above, that the addition of highly dispersed glass into a concrete composition suppresses alkali-silica reaction. It can be probably explained by the interaction of a free alkali with an active silica in the initial period of concrete hardening. To stimulate the process, it is also possible to use highly dispersed additives containing reactive amorphous silica. The effect of the additive will be greater, if it does not contain large quantities of Na+ ions. The influence of additives (dispersed silica gel, aerosil and ion-exchange modified glass) meeting the requirements on the ASR rate has been studied.
Dispersed silicate glass can be considered as a cationic ion-exchanger. This property can be used to obtain an H-glass – the glass, on the which surface Na+ - ions are replaced by H+-ions in the process of the ion-exchange reaction taking place in the solution of an acetic acid.
The results of experiments have shown, that the addition of the H-glass into a concrete composition helps to obtain concretes with a higher strength characteristics and to suppress alkali-silica reaction to some extend.
Chemical modification of the aggregate – ion-exchange elimination of Na+ ions from the glass surface - happened to be rather effective. It was found out that an expansion of glass concrete bars containing chemically modified glass did not exceed 0.05% when the glass particle size was less than 0.16 mm. But when glass aggregate with a greater particle size was used, the expansion exceeded the permissible limit in already 72 hours. The positive effect of the chemical modification of the glass aggregate consisted in the growth of strength characteristics of the glass concrete. The compressive strength of the chemically modified glass concrete was 1.5 greater than that of a typical glass concrete (the aggregate size was less than 0.16 mm). Since chemical modification allows increasing a glass concrete strength, optimum modification conditions have been developed – concentration of acid, time and treatment temperature, to obtain chemically modified glass with the required characteristics.
The addition of such dispersed additives as aerosil and silica gel effectively suppresses alkali-silica reaction. The alkali-silica reaction rate and the expansion of samples decrease with the increase of the additive content up to the total termination of the process.
Preliminary Treatment of Glass Cullet
As it was noted above, one of the methods of the ASR suppression is a process of transformation of anamorphous silica of the concrete aggregate surface into a less reactive form. Glass contains amorphous silicates which are thermodynamically unstable under normal conditions. Then it is possible to induce crystallization of glass, which results in formation of crystalline silicate compounds not participating in the alkali-silica reaction. To achieve this, we have carried out thermal treatment of glass cullet in the presence of various dispersed crystalline silicate materials. We supposed that such treatment may result in crystallization of glass at least only on its surface – that would be enough for obtaining an ASR-stable aggregate.
Thus, we have carried out thermal treatment of glass cullet in a drum furnace at 800oС, with the presence of various dispersed silicate materials. Then the glass cullet was used as an aggregate. The experiments have shown that the maximum expansion of the sample concrete blocks made with an addition of thermally modified glass does not exceed 0.1%, which is 7 times less than the expansion of the reference sample. The samples with a large fraction aggregate demonstrated a high strength. Addition of alkali into the concrete composition lowers its strength, but does not cause a considerable expansion.
Foamed Glass Gravel
Foamed glass gravel can also be used as an aggregate for concrete. It enables decreasing a total weight of a construction and reducing expenses. But the possibility of alkali-silica reaction in the case of a foamed glass aggregate should also be considered.
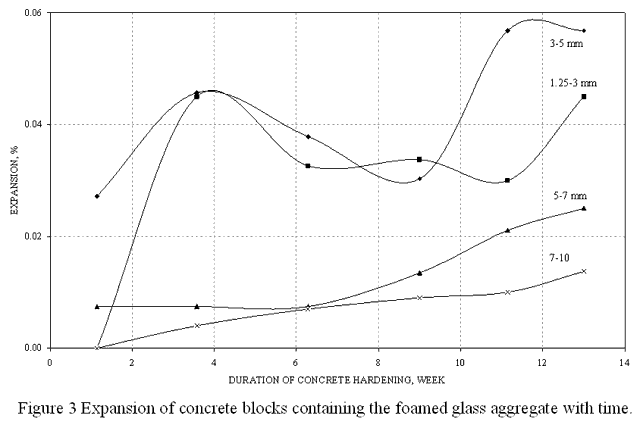
We have carried out a series of appropriate experiments. The results obtained – an expansion of concrete blocks containing the foamed glass aggregate of various granular sizes with time - are shown on Figure 3. It is evident, that the maximum expansion for all samples containing foamed glass does not exceed 0.1%. It means that the alkali-silica reaction is practically suppressed. The expansion of blocks decreases with an increase of a granular size of the aggregate.
So, the behavior of foamed glass in the concrete is similar to the behavior of thermally modified glass. It may be explained by the fact that the process of foamed glass production implies chemical treatment of the material in the presence of a heat carrier – quartz sand. Such treatment may result in crystallization of glass on the surface of granules and transformation of silicates into an inactive form.
CONCLUSIONS
Glass cullet can be used as an aggregate for concrete if the alkali-silica reaction is suppressed. Glass concrete with high quality and excellent strength characteristics can be obtained even from the cements with a high alkali content, but certain preventive measures should be taken. Two methods of ASR suppression have been suggested: 1) application of highly dispersed amorphous silicate additives, and 2) preliminary thermal treatment of glass cullet in the presence of crystalline silicates, inducing surface crystallization of glass particles.
The obtained results prove that glass can be used as an aggregate for producing concrete with a relatively high strength (up to 50 MPa) which will not deteriorate with time.
ACKNOWLEDGEMENTS
The authors would like to acknowledge the financial help provided by JCS Perm Production of Foamed Silicates for this project.
REFERENCES:
- LUDWIG, UDO. Einflusse auf die Alcali-Zuschlag-Reaction. Cem. and Concr. Res. 1976, №6. pp 765-772.
- MEYER, C, EGOSI, N, ANDELA, C. Concrete with waste glass as agregate // Recycling and Reuse of glass Cullet: Proceedings of International Symposium 19-20 March 2001, Dundee UK. pp 179–181.
- BYARS, E A, ZHU, H, MEYER, C. Use of glass for construction products: legislative and technical issues // Sustainable Waste Management: Proceedings of the International Symposium 9-11 September 2003, Dundee UK. pp 827–838.
- SUGIYAMA, M. The experiment on compression strength and freeze-thaw resistance of the concrete which mixed the tile clip // Recycling and Reuse of glass Cullet: Proceedings of International Symposium 19-20 March 2001, Dundee UK. pp 189–194.
- REMARQUE, W, HEINZ, D, SCHLEUSSER, C. Glass powder as a reactive addition for blast furnace cements // Recycling and Reuse of glass Cullet: Proceedings of International Symposium 19-20 March 2001, Dundee UK. pp 229–238.
- DHIR, R K, DYER, T D, TANG, M C. Expansion due to alkali-silica reaction (ASR) of glass cullet used in concrete // Sustainable Waste Management: Proceedings of the International Symposium 9-11 September 2003, Dundee UK. pp 751–760.
- MELAND, I, DAHL, P A. Recycling glass cullet as concrete aggregates, applicability and durability // Recycling and Reuse of glass Cullet: Proceedings of International Symposium 19-20 March 2001, Dundee UK. pp 167–177.
- DYER, T D, DHIR, R K. Use of glass cullet as a cement component in concrete // Recycling and Reuse of glass Cullet: Proceedings of International Symposium 19-20 March 2001, Dundee UK. pp 157–166.














